How does pH influence food? What role does it play in gourmet preserves and ready meals?
If you have been able to read the different articles that we offer you in our Blog, you will have seen that in many of them we mention pH. And the fact is that the acidity level of a food is crucial when it comes to subjecting it to heat treatment to preserve it longer, such as pasteurization or sterilization.
What is pH?
The acronym pH stands for hydrogen potential and indicates the degree of concentration of hydrogen ions in a food or any other type of solution. Surely you have also heard about the pH of gels or swimming pools. pH is used to determine the acidity level of a compound.
The pH scale ranges from 0 to 14. Being 1 the most acidic value, and 14 being the most basic, which is how products that are not very acidic are called. Those with a value of 7 are considered neutral, neither acidic nor basic, and those above 7 are alkaline or alkalizing.
The way to quantify the level of acidity is by the rule of less is more and more is less. That is, the more acidic a food is, the lower its pH value and vice versa.
The vast majority of foods are on a pH scale of 3 to 7 (except, for example, egg whites, which are above 7).
How can I distinguish between an acid and an alkaline product?
In general it is easy, you can distinguish them by their taste. Sour in the case of acidic foods and bitter in the case of alkaline ones.
How is pH measured?
The level of acidity can be measured in different ways. The simplest and most basic is using a strip of litmus paper, which you can buy in pharmacies. When you touch something with this paper, its color changes. If the substance is acidic, the paper turns red and if it turns blue it is because the substance is basic.
Normally, in Agrifood Analysis laboratories they use the pHmeter since iridescent paper is unreliable when it comes to measuring the pH in foods that contain various ingredients and textures because they can stain the paper and mask the result. In addition, the reading is done visually so they are not suitable for research either. In contrast, pHmeters have a much higher precision, sensitivity and accuracy.
If this is your case, that you take the readings using strips, we recommend that you have a pHmeter to more reliably assess the acidity level of your canned food and ready meals in order to carry out the most appropriate heat treatment to extend its useful life. with full guarantee.
And why is pH so important when preserving food?
It is so important because pH determines the time and temperature to which we must submit a food product to effectively eliminate the microbiological and enzymatic activity, ensuring the stability of the final food product.
As a general rule, longer times with low temperatures will kill the same amount of microorganisms as a high temperature and a short time, but they can also cause changes in the smell, color and taste of food in a more or less significant way.
Did you know that the pH of a food can be changed naturally?

Yes, you can increase the degree of acidity of your food product by adding more acidic ingredients, for example, more tomato if it already has it on the recipe or by adding vinegar or lemon.
Although in addition to acidity you will also add flavor, so it is only recommended in those cases in which this ingredient is part of the ready meal already or they do not alter its flavor especially.
The main function of lowering the pH of your preserve and, therefore, increasing its acidity, is to improve its preservation and/or allow a gentler and less aggressive heat treatment. And this is done because the risk in acidic foods of containing pathogenic bacteria is lower because the acidity hinders the growth of microorganisms.
There are bacteria that do not grow in an acid medium, including the dreaded Clostridium botulinum.
Most pathogenic microorganisms grow at a rather neutral pH, between 5 and 8. This does not mean that some pathogens cannot proliferate in foods with acidic pH, although to a lesser extent.
In the same way that the increase in acidity limits the growth of microorganisms, the action of enzymes and their resistance to temperature are also conditioned.
These microorganisms are more sensitive to heat treatment, so shorter cycles can be made or even at a lower temperature.
The effects of pH changes
Everything in this life has consequences and the modification of the degree of acidity of a food could not be less.
Colour
Reducing the pH of a preserve has a direct impact on its color, especially if it contains vegetables.
Texture
Proteins
Reducing the pH can cause variations in proteins by changing the texture of protein foods: meat, fish, dairy, etc.
Thickeners
Likewise, an acidic pH added to the high sterilization temperature, make the sauces thickened with flours or starches end up liquefying over time.
Sugars
Preserves with low pH and sugar, when sterilized, respond to heat by forming the so-called inverted sugar, which is sweeter and crystallizes less.
Remember that subjecting a gourmet preserve or ready meal to a heat treatment to extend its shelf life, may cause changes in the smell, color and flavor of food in a more or less significant way.
Technical and food consultancy
Throughout the article we have explained to you how the pH influences the organoleptic characteristics of your gourmet preserve or ready meal.
TERRA Food-Tech® technical team is aware of the importance of acidity in the preparation of different food products that our clients make. And it is no wonder, there are many aspects to take into account.
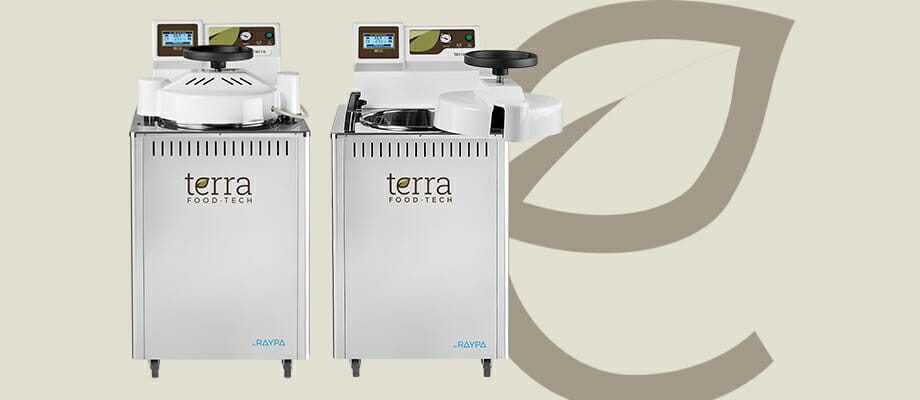
For this reason, our autoclaves contemplate these vicissitudes and have a series of tools whose main objective is to respect the organoleptic and nutritional characteristics of your gourmet preserve or ready meal. And they do it by incorporating:
- Direct control over the F0-P0 value in order to optimize the most appropriate combination of time-temperature for your specific production process.
- Heart temperature probe for samples with a perforator kit to be able to perfectly control the internal temperature of the product throughout the cycle.
- Continuous monitoring of all sterilization parameters. In case of failure or not reaching the expected results, the program is interrupted and generates a warning message.
Likewise, we put at your disposal our consultancy service, where our experts will guide you, from start to finish, in the preparation of your preserved food product. From solving problems in your production process to doubts regarding legislation, batch labeling and other technical issues.
For more information, do not hesitate to contact us, we will be happy to help you.