Did you know that the pH of food is essential for preservation and safety of gourmet preserved food and ready to eat meals?
The pH value indicates the hydrogen potential or potential of hydrogenions and it is used to determine the degree of alkalinity or acidity of a specific food or any other type of solution, based on the concentration of positive hydrogen ions contained in the compound.
The pH scale ranges from 0 to 14. A food is considered to be very acidic when its pH value is between 0 and 4, and alkaline or low acidity when its pH is above 4.5. Foods with a value between 4 and 4.5 are considered neutral or acidic.
The method for quantifying the acidity level is by the rule of less is more and more is less. That is, the more acidic a food is, the lower its pH value will be, and vice versa.
The pH reflects the composition of the food itself. Just like time, temperature, nutrients, water and salinity, pH also affects the shelf life of products. It is also a fundamental value when it comes to pasteurizing or sterilizing food, since depending on its degree of acidity or alkalinity, more or less microorganisms will proliferate.
How can we measure the pH of a preserved food or ready to eat dish?

Both gourmet preserved food and ready to eat meals combine different ingredients in the same container, which means there is a pH range. This is why it is so important to know the exact acidity level of a mixture. The pH of foods can be measured in two ways:
- Through a pH meter, a device that has a sensor bulb with two electrodes, one is calibrated and the other one is sensitive to H+ ions. When it is inserted into food, the potential difference between the electrodes is activated and the exact pH value appears on the digital display. It is the most accurate and quickest way.
- Using litmus paper strips. These are strips of paper impregnated with a mixture of indicators that, when they are immersed in foods, change their color depending on food acidity. The pH value is obtained by comparison with a color scale shown on the container. This is a homemade, inexpensive and rather less accurate way, not recommended for professional production of preserved foods and ready to eat meals.
When making pH measurements, it is important to take temperature into account, since this factor also affects the acidity level of a product. Usually, samples should be taken at room temperature (20ºC).
Did you know pH changes depending on the variety and ripeness of food? Yes, it does. For example, the acidity level of a golden apple is a bit different than a reineta apple, and it also depends on its ripeness. A very ripe food is less acidic than the same unripe food.
Food classification according to pH
As we already mentioned, the pH scale of foods ranges from 0 to 14, although most of them are between 1.5 and 10. However, we can also sort food products into different categories according to their food safety risks, since microorganisms grow mainly in less acidic environments.
Thus, if we take a pH of 4.5 as a reference value, a figure above which Clostridium botulinum cannot grow, we could classify foods as follows:
Low-acid foods or with low acidity level
When the pH value of a food is above 4.5, it is called a low-acid food or a food with low acidity level. This category includes most vegetables and meats. These are foods with higher risk, in which a large number of microorganisms can proliferate, like fungi, yeasts and pathogenic bacteria, such as Clostridium botulinum.
Acidic foods
These are foods with a pH value between 4 and 4.5, in which Clostridium botulinum does not germinate, but bacteria such as Bacillus coagulans or Bacillus licheniformis can grow.
High-acid foods
These are foods with a pH value equal to or lower than 4. Within this category we find citrus fruits, cereals, yogurts… Sporulates do not grow in very acidic foods, but molds, yeasts and acidophilic bacteria can develop.
It is worth mentioning that the most common foods are acidic or low acidic, since they are generally on a pH scale from 3 to 7.
Did you know you can distinguish an acidic food from an alkaline food by its taste? Acidic ingredients are known for their sour taste and alkaline ingredients for their bitter taste.
Importance of food pH in microorganism proliferation
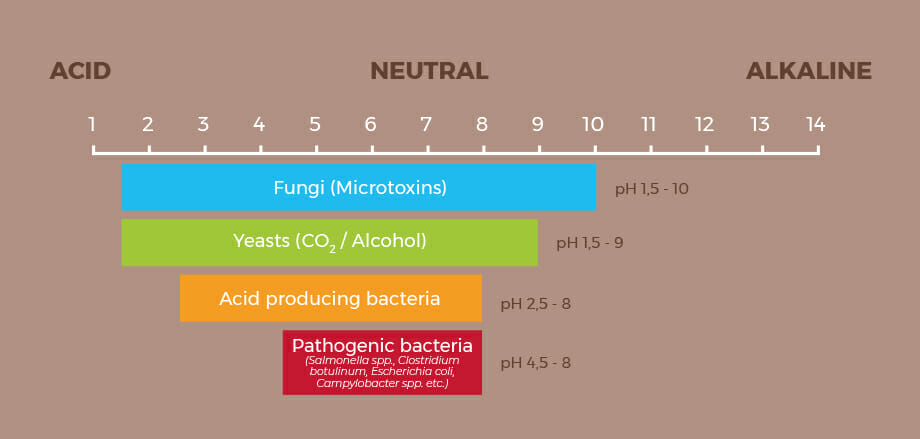
Therefore, as you have seen in food classification, pH determines what type of microorganisms can grow in them. The more acidic a food is, with low pH, the more difficult it is for microorganisms to survive and grow in it. The acidity of a product itself is both a means of preservation and a way of keeping food safe for consumption.
Development of microorganisms according to food pH
The different pH levels determine the type of chemical and biological reactions in a particular food. Below, we detail how different microorganisms evolve according to different food pH values:
Molds
Most molds grow in a pH range between 3.5 and 8.0, although they generally prefer an acidic pH.
Like some bacteria, molds can alter the pH of an unbuffered medium thanks to the substances they generate during their development.
Bacteria
In this group we find, among the most common cases, Listeria, Salmonella or Campylobacter. Almost all bacteria are neutrophilic, that is, they need a neutral pH to grow. However, there are also other bacteria that are capable of growing in very acidic pH, such as Staphylococcus aureus, Listeria monocytogenes or Salmonella spp.
Yeasts
Yeasts grow mostly in a range from 3.5 to 4.5, although it depends on presence of sugars, organic acids and other easily metabolized carbon sources. Yeasts tolerate acidic environments very well.
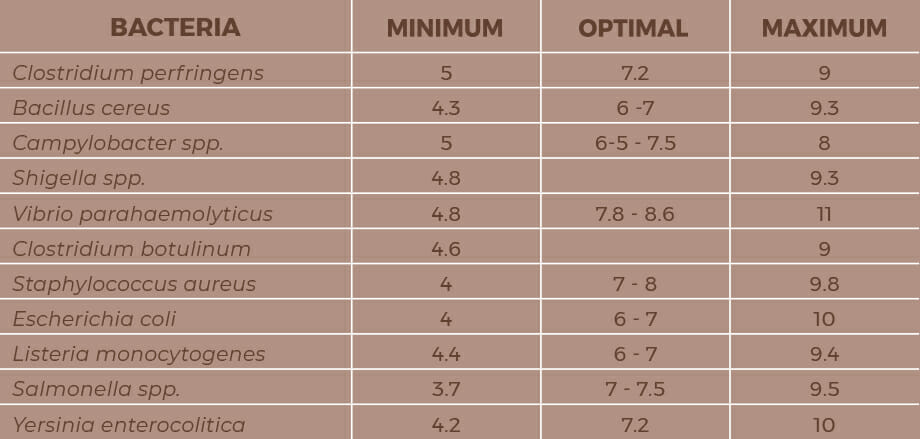
Importance of pH in pasteurization and/or sterilization of gourmet preserved foods and ready to eat meals
Food pH plays an important role when it comes to pasteurizing or sterilizing a gourmet preserved food or a ready to eat meal because it determines the time and temperature we must expose our product in order to eliminate microbiological and enzymatic activity.
Lower temperature during a longer time will eliminate the same amount of microorganisms as high temperature during a short time, but may also cause changes in smell, color and taste of foods in a more or less significant way.
Acidification or alteration of food pH

Food acidification consists of reducing, by natural or artificial means, food pH to prevent the development of pathogenic microorganisms. Lowering the pH of your preserved product means increasing its acidity and, therefore, improving its shelf life and allowing a gentler and less aggressive heat treatment to be applied.
Remember that most pathogenic microorganisms do not grow in acidic pH foods, although there are a few that can do.
When you increase the acidity of your preserved foods, you must take into account that, in addition to acidity, you will also add flavor. For this reason, we recommend that, when it comes to acidifying your canned food, you should add more acidic ingredients that are already part of the recipe itself, such as tomato, or ingredients that do not alter its flavor excessively, such as vinegar or lemon.
The main pH correctors include sodium carbonate, potassium, magnesium, citric and lactic acid, calcium acetate and fumaric acid.
We would like to remind you that adding acidulant substances does not guarantee food preservation for long periods of time. In order to ensure proper food preservation and meet sanitary standards, it is essential to sterilize or pasteurize your preserved foods or ready to eat meals.
pH chart of the most common foods
Below, we offer you a downloadable document with the pH values of the most common foods sorted in alphabetical order and by food type. We hope you find it very useful!
Food consulting regarding pH control of your preserved food or ready to eat meals
However, remember that pH of a preserved food or ready to eat meal does not depend on the individual ingredients, but on the resulting combination. Therefore, when sterilizing or pasteurizing your products, remember that you must know their overall pH level in order to apply the most appropriate time and temperature to always respect their organoleptic and nutritional characteristics. Because every aspect influences quality, taste, color, smell and texture of your packaged product.
At TERRA Food-Tech® we are aware of how difficult it can be to measure pH correctly. That is why we offer you our technical and agri-food consulting service. Our experts will guide you, from start to finish, in the preparation of your gourmet preserved foods or ready to eat meals.
For further information about our autoclaves and our services, do not hesitate to contact us, we will be pleased to help you.